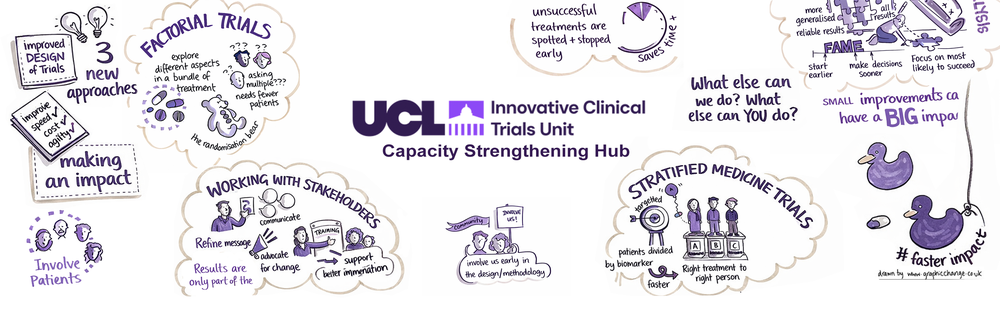
Welcome to the UCL Innovative Clinical Trials Unit Capacity Strengthening Hub
This Hub aims to provide resources on the design, conduct, analysis, and knowledge transfer and exchange for randomised controlled trials, observational studies, and meta-analyses.
Visit the UCL Innovative Clinical Trials Unit website for further information
Browse or search for resources on the design, conduct, analysis, and knowledge transfer and exchange for randomised controlled trials, observational studies, and meta-analyses.
A compilation of resources taken from the MRC CTU resources page, with a certificate available on completion.
Become a member of the MRC CTU at UCL Capacity Strengthening Hub today! You can opt-in to hear about upcoming events, new resources, and the latest news.

Help us improve the Hub by leaving some brief feedback